 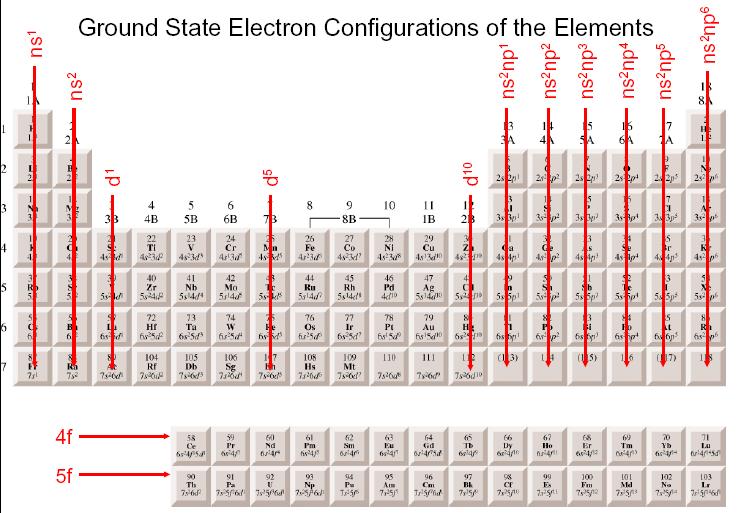 
Malleability and Ductility: Transition metals are generally malleable and ductile.The energy level differences between the d-orbitals contribute to the broad spectrum of colors observed. Colorful Compounds: Transition metal compounds often exhibit vibrant colors due to the absorption and emission of specific wavelengths of light by the d-electrons.This is a result of the presence of unpaired electrons in their d-orbitals. Paramagnetic Behavior: Many transition metals are paramagnetic, meaning they are attracted to an external magnetic field.The close packing of atoms in their crystal structures, along with their relatively large atomic masses, leads to their higher densities. High Density: Transition metals tend to have higher densities compared to other elements.The strong metallic bonding resulting from the overlapping of d-orbitals contributes to their high melting and boiling points. High Melting and Boiling Points: d block elements generally have higher melting and boiling points compared to s-block elements. 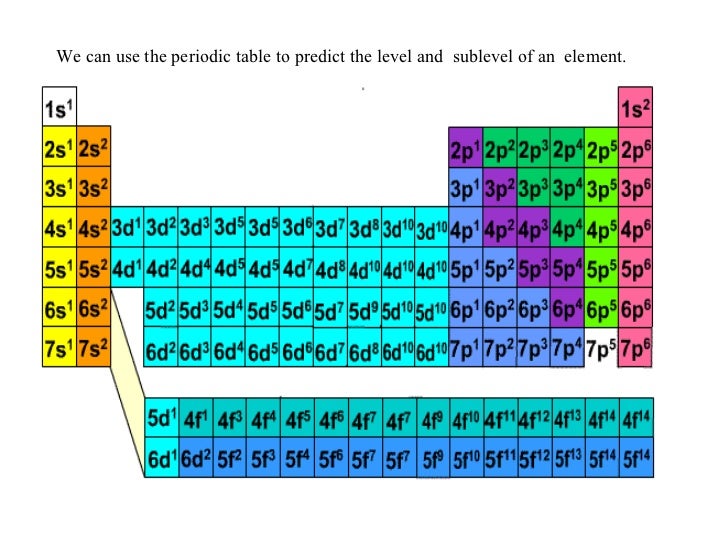
Their ability to undergo redox reactions and their versatile oxidation states make them effective in facilitating chemical reactions by providing an alternate pathway with lower activation energy. Catalytic Activity: Transition metals are excellent catalysts.For example, iron (Fe) can exist in the +2 or +3 oxidation states. They can readily lose or gain electrons to form ions with different charges. Variable Oxidation States: Transition metals exhibit multiple oxidation states due to the availability of their d-orbitals.These elements are less reactive than the s-block elements, but they are more reactive than the p-block elements.Īdvantages Of PW All India Test Series For JEE : Click to Check Properties of d block elements The d block elements are located in the middle of the periodic table.These elements are less reactive than the s-block elements, and they tend to form covalent compounds. The p-block elements are located on the right side of the periodic table, and they have valence electrons in the p-orbital.These elements are highly reactive and tend to form ionic compounds. The s-block elements are located on the left side of the periodic table, and they have valence electrons in the s-orbital.Their properties are intermediate between highly reactive metallic elements of the s-block, which are typically ionic compounds, and elements of the p-block, which are mostly covalent. JEE Main Syllabus Reduced : Click to Check Why d block Elements are Known as Transition Metals?ĭ block elements are known as transition elements because they exhibit transitional behavior between s-block and p-block elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
